The Zika Virus
Genetically modified mosquitoes can protect us from deadly diseases, writes chemistry professor Marc Zimmer.
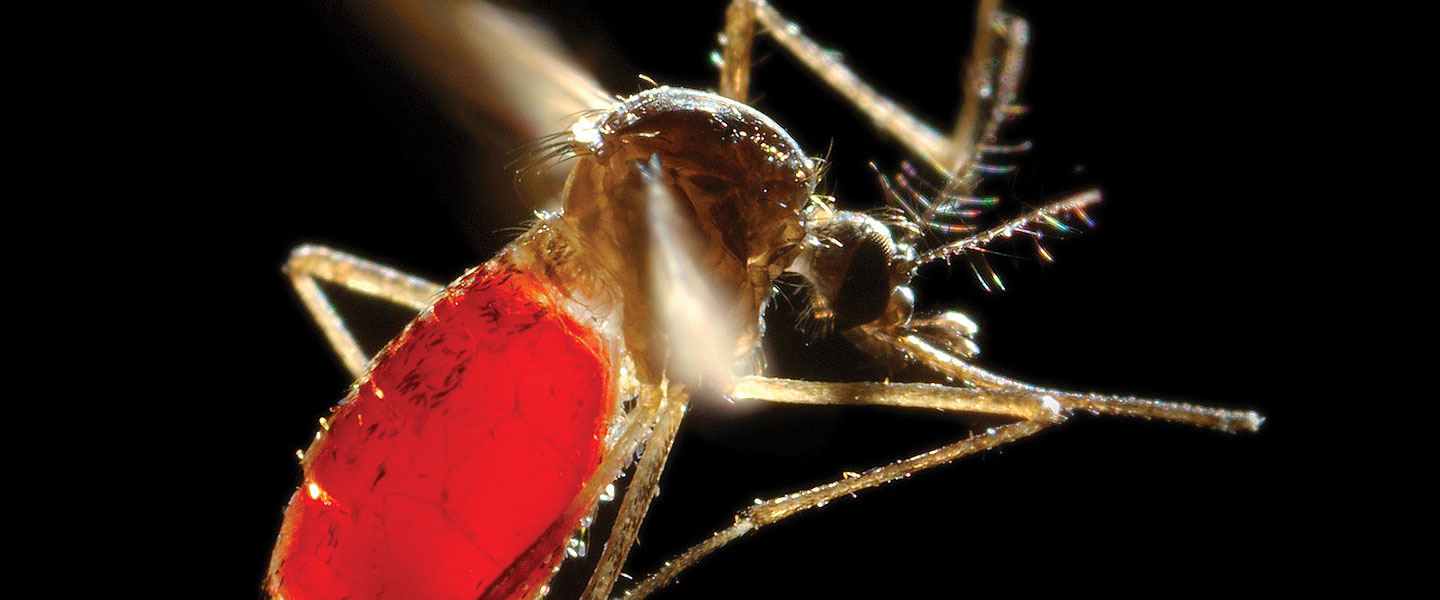
The world’s most dangerous animal is not a hippopotamus. It’s not a crocodile, or a lion, or a great white shark. It’s a mosquito.
These tiny critters are vectors of disease, passing devastating and deadly viruses—including Zika—from host to host. How can we stop them? One answer may surprise you: genetically modified mosquitoes.
Zika is not new, but for many decades it remained in the shadow of its older and more common sibling, the Dengue virus. Now, with the recent outbreak of Zika—and its association with a birth defect known as microcephaly—we will have to use everything we have learned about Dengue to control it. That includes releasing genetically modified mosquitoes into impacted areas.
The Zika virus got off to a slow start. The symptoms, which include fever, rash, joint pain and conjunctivitis (pink eye), are typically mild—so mild, in fact, they often go unnoticed. In the 70 years since Zika was first identified in Uganda, less than five cases have been confirmed in that country. However, many people in Uganda have Zika antibodies in their blood, meaning that they were infected but never sick enough to seek treatment. Zika antibodies have also been found in India, Vietnam, Thailand, Indonesia, Malaysia and the Philippines. Yet between 1947 and 2006, there were just 14 confirmed cases worldwide.
In 2007, something changed. Perhaps the Zika virus mutated; suddenly, it was on the move. That year, 50 people were infected on the Micronesian island of Yap. Six years later, thousands fell victim to the virus in French Polynesia, about 5,000 miles away. And now more than 1.5 million people in Brazil appear to have Zika. Not only is the virus spreading more efficiently, but, more alarming, it is also causing microcephaly, a birth defect in which babies’ heads are unusually small and their brains do not develop properly.
Zika is most commonly transmitted by the Aedes aegypti, also known as the “yellow fever mosquito.” This is the same pest primarily responsible for spreading the Dengue virus, characterized by fever, pain in the eyes, a rash, and oral, vaginal and intestinal bleeding. Although Aedes originated in Africa, they are now found in tropical and subtropical areas throughout the world; 40 percent of the world’s population is threatened by Dengue, and it’s a leading cause of illness and death in the tropics and subtropics. Like Zika, there is no treatment or vaccine for Dengue fever.
That these two viruses are primarily transmitted by the same type of mosquito is both good news and bad. On the plus side, in our efforts to fight Dengue, we have learned a lot about controlling Aedes populations that we can now apply to Zika-affected areas.
On the other hand, these are stealthy little creatures. They are smaller and quieter than the mosquitoes typically found in the United States; there is no annoying buzz to warn potential victims. They feed for one-to-two hour periods in the morning and late afternoon, so mosquito netting around bedding provides little protection. They thrive in urban environments; their ideal habitat is close to human homes where they can avoid pesticide spraying. And although Aedes travel less than a mile in their lifetime, humans do travel, and Aedes can pick up viruses from humans and pass them to their offspring.
Controlling and limiting the habitat of these mosquitoes is extremely difficult, as they can lay eggs in a single drop of water. This has led to severe restrictions in several countries: in Singapore, a homeowner can be fined for having Aedes breeding sites, such as a glass of water in the garden; in Malaysia, the home of anyone who contracts Dengue fever and the homes of their neighbors have to be sprayed—inside and out—with pesticides. These insecticides can be harmful to humans, particularly those exposed to them for long periods of time (such as the people doing the spraying) and children. Over time, populations of mosquitoes can also become resistant to pesticides.
Enter the genetically modified fluorescent mosquito.
Created by British biotech company Oxitec, these Aedes mosquitoes are designed with a self-limiting gene that causes their offspring to die in the larval stage. Oxitec breeds these modified mosquitoes by feeding them an antidote that turns the destructive gene off in captivity. The mosquitoes are then sorted according to gender, and, because only pregnant females bite humans (males eat nectar; females need blood to help their eggs develop), the females are killed before the males are released. The released males spend their lives searching for wild females in the area where they were freed. They mate, but all the offspring they produce will have the lethal gene and die.
If enough modified males are released, the entire Aedes population will collapse.